As bipartisan momentum to hold Big Pharma accountable and take action on out-of-control drug prices continues to build, the U.S. Senate Committee on Finance is scheduled to question the executives of seven major pharmaceutical manufacturers on February 26, 2019.
In advance of the hearing, the second held by the committee on drug pricing this year, the Campaign for Sustainable Rx Pricing (CSRxP) is rolling out a new blog series on what lawmakers can expect to hear from these seven pharma CEOs – and why their rhetoric rings hollow when measured against their companies’ track record.
We’re taking a look at the unkept pledges, proclamations and promises made by Big Pharma that lawmakers should be prepared to confront with a “dose of reality.”
In this dose of reality, we’ll see how the rhetoric of Big Pharma CEOs matches up with reality (“the hard medicine”).
ABBVIE CEO RICHARD GONZALEZ:
Richard’s Rhetoric:
- “We Make Decisions Based On Our Deep Caring And Compassion For People, Delivering A Lasting Impact To Our Patients, Their Families, Our Employees, And The Community.” (“About AbbVie,” AbbVie, Accessed 2/7/19)
- “We Strive To Always Do The Right Thing. With Uncompromising Integrity At The Heart Of Everything We Do, We Pursue The Highest Standards In Quality, Compliance, Safety, And Performance.” (“About AbbVie,” AbbVie, Accessed 2/7/19)
HARD MEDICINE:
- AbbVie Has Raised The Price Of Its Best-Selling Drug, Humira, By 100 Percent From 2012-2018. (Danny Hakim, “Humira’s Best-Selling Drug Formula: Start At A High Price. Go Higher.” The New York Times, 1/6/18)
- Originally Set To Lose Patent Protection In 2014, AbbVie Has More Than Doubled The Span Under Which Its Blockbuster Humira Is Covered, With The Latest Expiration Now In 2034. “The company listed 22 patents for various diseases or methods of treatment, 14 on the drug’s formulation, 24 on its manufacturing practices, and 15 ‘other’ patents. The latest expiration date is 2034 – providing more than double the protection span a drug such as Humira might normally expect.” (Cynthia Koons, “This Shield Of Patents Protects The World’s Best-Selling Drug,” Bloomberg Businessweek, 9/7/17)
- “247 Patent Applications Have Been Filed On Humira In The U.S. With The Aim Of Delaying Competition For 39 Years.” (“Overpatented, Overpriced: Special Humira Edition,” I-MAK, Accessed 2/7/19)
- In September 2018, AbbVie’s Chief Financial Officer Bill Chase Boasted About Extending Humira’s U.S. Monopoly Saying, “You’ve Seen Us Execute Very Nicely With Our Legal Strategy And The Settlements Around The U.S. Events To Delay The Onset Of [Loss Of Humira’s Exclusivity] Into The 2022-2023 Time Period.” “Cheaper versions of Humira are hitting European markets next month, and a top AbbVie executive is celebrating two separate deals that would prevent that from happening in the U.S. – where Humira’s annual costs are as much as a high-end car – for five years.” (Bob Herman, “AbbVie Boasts Of Extending Humira’s U.S. Monopoly,” Axios, 9/12/18)
ASTRAZENECA CEO PASCAL SORIOT:
Pascal’s Pledges:
- “We Follow The Science. We Put Patients First. We Play To Win. We Do The Right Thing. We Are Entrepreneurial.” (“Our Company,” AstraZeneca, Accessed 1/16/19)
- “We Aim To Improve Access To Healthcare And Deliver Our Medicines, Services And Education To Patients Around The World By Tailoring Our Programmes To The Communities They Will Serve. In Some Cases, This Means Looking At Pricing; In Others It Involves Overcoming Other Barriers To Healthcare.” (“Our Company,” AstraZeneca, Accessed 1/16/19)
- “Fundamentally, This Industry Can Only Create Shareholder Value By Creating Value For Patients. It’s Pretty Simple. If You Don’t Come Up With Medicines That Help Patients, There Is No Way You Can Have A Sustainable Business Creating Value For Shareholders.” (Matthew Herper, “AstraZeneca CEO On His Predecessors, Rivals, And Pfizer,” Forbes, 6/3/14)
HARD MEDICINE:
- As AstraZeneca Faced Generic Competition To Its High Cholesterol Drug Crestor, Its “Price Was Increased Several Times Before The Generic Came Out … Including By About 15 Percent Right Before.” “AstraZeneca’s AZN, -0.08% drug Crestor, another of the drugs featured in the report, is a popular but expensive drug that treats high cholesterol. In 2016, when the drug first got a new generic rival, the branded product cost about $300 a month without insurance coverage. The price was increased several times before the generic came out … including by about 15% right before. (AstraZeneca said it could not comment because it was not involved in the study.)” (Emma Court, “Big Pharma Games The System To Make Generic Drugs More Expensive,” MarketWatch, 8/3/18)
-
- AstraZeneca’s Pricing Strategy Served To Create “A New, Higher Baseline Price When The Generic Hits The Market.”(Tori Marsh, “Prices For Brand Drugs Spike Before A Generic Is Released. Here’s Why.,” GoodRx, 7/27/18)
- After Increasing Drug Prices By As Much As Nine Percent, On A 2018 Earnings Call, Soriot Insisted The Company Was “Sensitive” To Drug Pricing Concerns And Said It Had Raised “Wholesale Prices Earlier [That] Year By ‘Very, Very Modest’ Amounts.” “During an earnings conference call, the AstraZeneca chief executive disclosed the company would not raise prices in the U.S. for the rest of year. Other drug makers have taken the same step in response to pressure from the Trump administration, but he insisted this was ‘our plan … all along’ … He maintained AstraZeneca was sensitive to the problem by raising wholesale prices earlier this year by ‘very, very modest’ amounts, ‘between 1 and 3 percent’ which, he said, was ‘in line with inflation.’” (Ed Silverman, “When Modest Is Actually Excessive: AstraZeneca Spins Its Price Hikes,” STAT News, 7/26/18)
BRISTOL MYERS SQUIBB CEO GIOVANNI CAFORIO:
Giovanni’s Guarantees:
- “Each Day, Our Employees Around The World Work Together For Patients – It Drives Everything We Do.” (“About Us,” Bristol-Myers Squibb, Accessed 2/7/19)
- “As Global Citizens, We Work Sustainably, Responsibly And Seek To Give Back.” (“About Us,” Bristol-Myers Squibb, Accessed 2/7/19)
HARD MEDICINE:
- In 2016, Bristol-Myers Squibb Agreed To A $19.5 Million Settlement Over Claims It “Improperly” Promoted Its Antipsychotic, Abilify, By “Minimizing Safety Risks And Overstating The Findings Of Scientific Studies That Were Used To Tout The Medication … However, The Drug Maker Did Not Admit To Any Wrongdoing.” (Ed Silverman, “Bristol-Myers Pays $19.5 Million To Settle Illegal Marketing Charges,” STAT News, 12/8/16)
- On The Same Day Bristol-Myers Squibb Announced Its Intention To Acquire Celgene, “Which Has Routinely Increased The Prices Of Its Top-Selling Drugs,” Celgene Hiked The Price Of The Dose Of Cancer Drug “Revlimid By 3.5 Percent To $719.82,” Compared With “$247.28 At The End Of 2007.” “On the same day Celgene Corp. was announcing that it would be acquired by Bristol-Myers Squibb Co. in the biggest pharma deal ever, the company was also raising the price of its blockbuster cancer drug. The Summit, New Jersey-based biotechnology company, which has routinely increased the prices of its top-selling drugs, boosted the price of a 10-milligram dose of Revlimid by 3.5 percent to $719.82 effective Jan. 3, according to price data compiled by Bloomberg Intelligence and First Databank. Cancer patients need many doses of Revlimid a year, and the overall cost can approach $200,000. The same dose cost $247.28 at the end of 2007.” (Rebecca Spalding, “Celgene Boosted The Price Of Its Top Cancer Drug On The Same Day Of Mega-Deal,” Bloomberg, 1/4/19)
- Lawmakers Have Raised Concerns The Deal Would “Stifle Competition, Particularly In Cancer, Causing Drug Prices To Rise And Hurting Consumers.” (Arlene Weintraub, “Congressmen To Regulators: BMS-Celgene Merger Will Stifle Competition And Raise Drug Prices,” FiercePharma, 1/15/19)
- In Addition To “Constructing An Almost Impenetrable Fortress Of Patents And Grants Of Market Exclusivity Around Revlimid,” Celgene Hiked Revlimid’s Price By Thousands Of Dollars A Month. “When Celgene Corp. first started marketing the drug Revlimid to treat multiple myeloma in 2006, the price was $6,195 for 21 capsules, a month’s supply. By the time David Mitchell started taking Revlimid in November 2010, Celgene had bumped the price up to about $8,000 a month. When he took his last month’s worth of pills in April 2016, the sticker price had reached $10,691. By last March, the list price had reached $16,691 …. Celgene has kept generic competition at bay by constructing an almost impenetrable fortress of patents and grants of market exclusivity around Revlimid, and its sister drug Thalomid, while also taking steps to ensure that generic competitors can’t get their hands on enough of the drugs to develop viable alternatives.” (Alison Kodjak, “How A Drugmaker Gamed The System To Keep Generic Competition Away,” NPR, 5/17/18)
- Patents And Exclusivity Designations On Revlimid Will Impede Generic Competitors’ Market Entry Until At Least 2022. “When Bristol-Myers Squibb picked up Celgene in one of biopharma’s biggest deals ever last month, investors tagged one big risk: Celgene’s megablockbuster Revlimid faced patent challenges that could take a big bite out of sales … The patent office’s decision should position Celgene and BMS to strike a favorable settlement with Dr. Reddy’s Laboratories, Credit Suisse Analyst Vamil Divan figures. Celgene has already settled with Natco Pharma in a deal that allows a limited generic launch in March 2022, he pointed out.” (Eric Sagonowsky, “BMS Can Breathe Easier Now: Celgene’s Revlimid Has Escaped A Big Patent Challenge,” FiercePharma, 2/12/19)
JOHNSON & JOHNSON CEO ALEX GORSKY:
Alex’s Assurances:
- “We Believe Our First Responsibility Is To The Patients, Doctors And Nurses, To Mothers And Fathers And All Others Who Use Our Products And Services. In Meeting Their Needs Everything We Do Must Be Of High Quality. We Must Constantly Strive To Provide Value, Reduce Our Costs And Maintain Reasonable Prices.” (“Our Credo,” Johnson & Johnson, Accessed 1/19/18)
- “We Are Responsible To The Communities In Which We Live And Work And To The World Community As Well. We Must Help People Be Healthier By Supporting Better Access And Care In More Places Around The World.” (“Our Credo,” Johnson & Johnson, Accessed 1/19/18)
- “If We Don’t [Price Drugs Reasonably And Be Transparent] As An Industry, I Think There Will Be Other Alternatives That Will Be More Onerous For Us.” “As politicians hammer drug prices, Johnson & Johnson CEO Alex Gorsky suggested companies need to police themselves. At the conference, Mr. Gorsky told investors that drug companies should price drugs reasonably and be transparent. ‘If we don’t do this as an industry, I think there will be other alternatives that will be more onerous for us,’ Mr. Gorsky says. Some drugmakers pulled back from price increases in mid-2018 amid heightened political scrutiny, but prices went up for many drugs at the start of 2019.” (Peter Loftus & Anna Wilde Mathews, “Health-Care CEOs Outline Strategies At J.P. Morgan Conference,” The Wall Street Journal, 1/9/19)
HARD MEDICINE:
- Since 2012, Johnson & Johnson Has Raised The Price Of Best Selling Drug Xarelto, A Drug Used To Treat Blood Clots, By 87 Percent. (“Overpatented, Overpriced: How Excessive Pharmaceutical Patenting Is Extending Monopolies And Driving Up Drug Prices,” I-MAK, 8/2/18)
- In January 2019, “Johnson & Johnson Raised U.S. Prices On Around Two Dozen Prescription Drug,” With Most Of The Increases “Between 6 Percent And 7 Percent.” (Michael Erman, “J&J Raises U.S. Prices On Around Two Dozen Drugs,” Reuters, 1/10/19)
- In 2018, Janssen, A Johnson & Johnson Pharmaceutical Subsidiary And Its Partner, Backed Away From Plans To Triple The Cost Of A Blood Cancer Drug Only After Significant Reproach From Oncologists.(Carolyn Y. Johnson, “Science Hinted That Cancer Patients Could Take Less Of A $148,000-A-Year Drug. Its Maker Tripled The Price Of A Pill.,” The Washington Post, 4/18/18)
MERCK CEO KENNETH FRAZIER:
Kenneth’s Commitments:
- “Merck Is Working To Save And Improve Lives Around The World By Inventing Breakthrough Medicines And Vaccines – And Ensuring They Are Available And Affordable To Patients Who Need Them.” (“Our Commitment To Access And Affordability,” Merck & Co., Accessed 2/11/19)
- “We Have A Long History Of Responsibly Pricing Our Medicines And Vaccines To Reflect Their Value To Patients, Payers And Society.” (“Our Commitment To Access And Affordability,” Merck & Co., Accessed 2/11/19)
HARD MEDICINE:
- From 2012 To 2017, Merck & Co. Raised The Price Of Its Drug To Treat High Cholesterol, Zetia, By 114 Percent. (“Manufactured Crisis,” S. Senate HSGAC, 3/27/18)
- In July 2018, Merck Said It Would “Lower Prices On Several Drugs By 10 Percent Or More,” However, The Drugs Impacted Were “Minor Products” With Dipping Sales Or That “Had Lost Their Patent Protection And Are Available … As Low-Cost Generics.” (Katie Thomas, “Merck Is Lowering Drug Prices. There’s A Catch.,” The New York Times, 7/19/18)
- The Drugs Receiving Cuts “Add[ed] Up To Less Than 0.1 Percent Of Merck’s Total Sales.” (Peter Loftus, “Merck To Limit Drug-Price Increases, Cut Some Prices,” The Wall Street Journal, 7/19/18)
- Merck’s List Prices Increased Nearly Seven Percent In 2017 And “Before That, Merck’s Average Annual List Prices Had Increased By At Least Nine Percent Since 2011.” (Peter Loftus, “Merck To Limit Drug-Price Increases, Cut Some Prices,” The Wall Street Journal, 7/19/18)
- “In Every Year After 2010, Merck Said Its Average List Price Hikes Over Its Entire Drug Portfolio Was More Than 9 Percent, Peaking At 10.5 Percent In 2014.” (Bill Berkrot, “Merck Reveals Seven Years Of Its U.S. Drug Price Increase History,” Reuters, 1/27/17)
PFIZER CEO ALBERT BOURLA:
Albert’s Assertions:
- “We Believe The Best Means To Address Affordability Of Medicines Is To Reduce The Growing Out-Of-Pocket Costs That Consumers Are Facing Due To High Deductibles And Co-Insurance, And Ensure That Patients Receive The Benefit Of Rebates At The Pharmacy Counter.” (Press Release, “Pfizer Provides Transparency On Drug Prices In The U.S. 90 Percent Of Company’s Prices Will Remain Unchanged,” Pfizer, Accessed 1/17/19)
HARD MEDICINE:
- Since 2012, Pfizer Has Hiked The Price Of Best Selling Drug, Lyrica, To Treat Pain, By 163 Percent.(“Overpatented, Overpriced: Special Humira Edition,” I-MAK, Accessed 2/7/19)
- From 2012 To 2017, Pfizer Raised The Price Of Its Heart Drug Nitrostat By 477 Percent. (“Manufactured Crisis,”U.S. Senate HSGAC, 3/27/18)
- Before A Generic Of The Heart Drug Nitrostat Came On Market, Pfizer Increased The Price “By About 56 Percent Overnight – From Around $0.80 To $1.25 Per Tablet” – Pushing The Generic Drugs Cost To 25 Percent “More Expensive Than Nitrostat Had Been Before Its Price Jump.” “Just before a generic for Nitrostat was released, the price per tablet of Nitrostat increased by about 56% overnight – from around $0.80 to $1.25 per tablet. A year later, generic nitroglycerin came on the market for around $1.00 per tablet, 25% more expensive than Nitrostat had been before its price jump.” (Tori Marsh, “Prices For Brand Drugs Spike Before A Generic Is Released. Here’s Why.,” GoodRx, 7/27/18)
- In July 2018, Pfizer Raised Prices On More Than 40 Prescriptions, “In Most Cases [By] More Than Nine Percent – Well Above The Rate Of Inflation In The US, Which [Was] Running At About Two Percent.” (David Crow, “US Drugmaker Pfizer Lifts Price Of Viagra And 100 Other Products,” Financial Times, 7/2/18)
- The July Increase Alone Raised The Price Of Chantix, A Smoking-Cessation Pill, Seven Percent To $429 For A Bottle Of 56 Tablets.“On July 1, the list price of a bottle of 56 tablets rose 7% to $429, after a 9.4% increase in January, according to Elsevier’s drug-price database.” (Jonathan D. Rockoff, “Pfizer Raises Prices For Dozens Of Drugs,” The Wall Street Journal, 7/2/18)
- In January 2018, “Pfizer, The Largest Standalone Drugmaker In The US, Raised The Average Wholesale Price Of 148 Drugs By A Mean Average Of 8.8 Percent.” “Pfizer, the largest standalone drugmaker in the US, raised the average wholesale price of 148 drugs by a mean average of 8.8 percent, according to data seen by the Financial Times. The list included several of its best-known medicines such as Viagra, the erectile dysfunction treatment, and Lyrica for nerve pain.” (David Crow, “Cost Of Viagra Soars As Hundreds Of Drug Prices Rise In US,” Financial Times, 1/4/18)
SANOFI CEO OLIVIER BRANDICOURT:
Olivier’s Overtures:
- “We Want Everyone – Including Patients, Providers, Payers, PBMs, Policy Makers, Regulators And Our Shareholders – To Understand Why We Set Prices As We Do, And To Reaffirm To Them Our Commitment To The Principles Of Access, Affordability And Innovation.” (Olivier Brandicourt, “Sanofi Pricing Principles For The U.S.,”Sanofi, Accessed 1/17/19)
- “We Continue To Believe Our Industry Has A Key Role To Play In Delivering Better Outcomes For Patients With Innovative Medicines That Are Cost-Effective For The Overall System.” (Olivier Brandicourt, “Sanofi Pricing Principles For The U.S.,” Sanofi, Accessed 1/17/19)
- Sanofi Said It Would Limit Its Price Increases To Medical Inflation And Would Provide “Information Publicly To Demonstrate The Rationale” Of Increases Above The Rate Of Medical Inflation. (Olivier Brandicourt, “Sanofi Pricing Principles For The U.S.,” Sanofi, Accessed 1/17/19)
HARD MEDICINE:
- Since 2012, Sanofi Has Hiked The Price Of Best Selling Insulin, Lantus By 114%. (“Overpatented, Overpriced: Lantus,” I-MAK, Accessed 2/7/19)
- “… Sanofi’s Popular Insulin Brand Lantus Was $35 A Vial When It Was Introduced In 2001; It’s Now $270,” A 671% Increase. (Tiffany Stanley, “Life, Death And Insulin,” The Washington Post, 1/7/19)
- According To The Initiative for Medicines, Access and Knowledge (I-MAK), “Lantus Is … Highly Overpatented” For The Purpose Of Being Able To “Preserve And Extend Its Ability To Keep Competition At Bay While Hiking Prices.” “Lantus is also highly overpatented. Though Sanofi’s primary patents on Lantus expired in 2015, the company has filed 70 secondary patent applications in the U.S. – 95 percent of its total – since the drug was first approved and put on the market in 2000. If granted, these additional patents would give Sanofi monopoly protection for up to 37 more years – almost double the duration provided under U.S. law. Why would a pharmaceutical company file so many patents after a drug is already on the market? Quite simply to preserve and extend its ability to keep competition at bay while hiking prices.” (Tahir Amin, “Patent Abuse Is Driving Up Drug Prices. Just Look At Lantus,” STAT, 12/7/18)
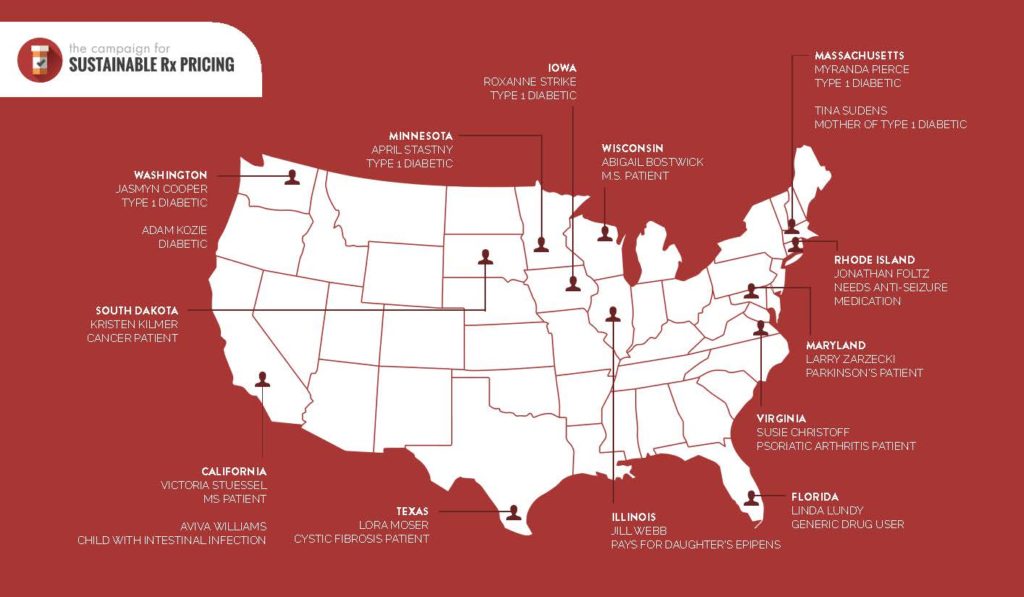
The truth is, Big Pharma’s price gouging is impacting patients from coast to coast – who are struggling to afford the medications they need to survive.
Click the below map the read more about their stories, and the impact out-of-control drug prices are truly having on every day Americans: